Tribute to Tom Hannon, written by Howard Samuels
It is with deep sadness that I share news of the passing of our dear friend, Tom Hannon. Tom was a cherished presence in all our lives – known for his warmth, kindness, and unwavering support for those around him. His sense of humor, generous spirit, and ability to brighten any room will be profoundly missed.
Tom was deeply committed to helping others through his involvement with the Advanced Bionics Bionic Ear Association, where he offered invaluable guidance and support to those navigating their hearing journeys. His dedication manifested itself in the cochlearimplantHELP website, which he co-founded and maintained as a resource for countless individuals and families seeking advice and encouragement. Tom particularly enjoyed employing his professional investigative skills to research details of advances in the cochlear implants. His attention to detail and careful maintenance of the comparison chart has benefited tens of thousands of individuals in their search to go beyond the glossy marketing brochures and to make informed decisions about their individual hearing journeys. Tom’s passion for empowering others through these communities left a lasting impact, and his contributions will continue to benefit many for years to come.

MED-EL and Starkey Announce Collaboration to Develop New Bluetooth Experience
Global Hearing Technology Leaders Celebrate Next Step in Innovation at CI2024
July 10, 2024 – DURHAM, NC – Global hearing implant leader MED-EL and leading hearing innovator in technology Starkey announced today a collaboration that will enable an all-new Bluetooth listening experience for people with a MED-EL cochlear implant (CI) and Starkey products. The announcement was made at CI2024, the 17th International Conference on Cochlear Implants and Other Implantable Technologies, in Vancouver.
The collaboration marks the beginning of an enhanced Bluetooth experience between MED-EL CIs and Starkey hearing aids. This allows users to take advantage of all streaming technology that Bluetooth offers.
“Last year, from the podium at my first ACIA meeting since joining MED-EL, I promised MED-EL would ask our customers more questions and listen to the answers. By announcing this exciting collaboration with Starkey today, we expect to co-develop solutions to enhance your MED-EL patient’s binaural hearing experience. Just like you asked,” said John Sparacio, President and CEO, MED-EL USA.
“Today marks the beginning of two global technology leaders coming together to help more people hear their very best. Partnering with MED-EL continues Starkey’s vision to always push the edge of what’s possible,” said Brandon Sawalich, President and CEO of Starkey.
The new technology will expand Bluetooth connectivity solutions for people using bimodal hearing technology.
About Starkey
Starkey is a privately held, global hearing technology company headquartered in Eden Prairie, Minnesota. Owned by Bill Austin since 1967, Starkey is known for its innovative design, development and distribution of comprehensive digital hearing systems. Led by President and CEO Brandon Sawalich, Starkey has more than 5,000 employees, operates 29 facilities and does business in more than 100 markets worldwide. Learn more at starkey.com.
About MED-EL
MED-EL Medical Electronics, a leader in implantable hearing solutions, is driven by a mission to overcome hearing loss as a barrier to communication and quality of life. The Austrian-based, privately owned business was co-founded by industry pioneers Ingeborg and Erwin Hochmair, whose ground-breaking research led to the development of the world’s first micro-electronic multi-channel cochlear implant (CI), which was successfully implanted in 1977 and was the basis for what is known as the modern CI today. This laid the foundation for the successful growth of the company in 1990, when they hired their first employees. To date, MED-EL has more than 2,700 employees from around 80 nations and 30 locations worldwide.
The company offers the widest range of implantable and non-implantable solutions to treat all types of hearing loss, enabling people in 136 countries enjoy the gift of hearing with the help of a MED-EL device. MED-EL’s hearing solutions include cochlear and middle ear implant systems, a combined electric acoustic stimulation hearing implant system, as well as surgical and non-surgical bone conduction devices. www.medel.com
# # #
MEDIA CONTACT:
FOR MED-EL
Rebecca Novak Tibbitt
FOR STARKEY
Jonell Lundquist
MED-EL Music Festival
MED-EL’s Sound Sensation Music Festival Celebrates International Artists with Hearing Loss
October 6, 2022 – (Durham, NC) – MED-EL, a global leader in implantable hearing solutions, has been passionate about connecting people with hearing loss to the magic of music since pioneering the cochlear implant in 1977. This year, between October 6-8, MED-EL will bring music alive at their Sound Sensation Festival. Singers and musicians from around the world who are deaf and hard of hearing and rely on MED-EL technology to hear will perform alongside renowned artists including a Vienna Philharmonic ensemble, violinist Yury Revich, and Eurovision winner Conchita Wurst, demonstrating the power of music in bringing people together.
To many, enjoying the multifaceted aspects of music is an integral part of life. Giving musicians with hearing loss a stage aims to inspire cochlear implant users around the globe. The virtual festival is for everyone who defies expectations with their hard work and natural talent to make world-class music.
“When people lose their hearing, they tell us that they miss the sound of family and friends, birds and music. Many of our incredible users go on to enjoy and play music again. Our festival is a celebration of their love for music and life. We see so many MED-EL users accomplish greatness in a multidimensional world of sound and music and strongly believe the world should see so too,” said Marcus Schmidt, Director of Corporate Marketing at MED-EL.
Organized and hosted by MED-EL, the virtual event will be the world’s biggest music festival the hearing implant community has seen. It will bring together individuals with hearing loss, their families, and ENT professionals as part of a series of regional and global online events. Small-scale concerts championed by hearing implant users, workshops, a symposium for hearing professionals will occur, followed by a live Grand Finale that will take place on October 8.
The three-day festival will feature a comprehensive program for everyone with a love for music. Viewers interested in listening to music and musical rehab can experience:
• Interactive workshops for parents, implant users and rehabilitation specialists
• Short daily concerts featuring musical performances from countries across the world
• A “Best Of” event showcasing a wide variety of MED-EL users’ musical activities
The entire festival will be broadcast globally and will be available on demand. For more information, please visit the event page here.
Market Research for US CI Users
A market research company is offering a $25 e-gift card to participate in a project on the topic of choosing cochlear implants and experience using them. Below is a link to a short survey. If you meet the criteria, then you may be sent an invitation to a 10-minute survey. Those who complete the 10-minute survey will receive a $25 e-gift card. It’s open to US residents with any brand cochlear implant, but there are limited spots. Click here to see if you qualify, and please feel free to share the link!
If you have any technical issues you can email CI_User_Survey@intellisurvey.com for help.
Comparison Chart Update
A new comparison chart is available with the latest offerings from Advanced Bionics, Cochlear, and MED-EL!
Advanced Bionics Naída CI M90 Review
David Ryan and Howard Samuels

First Impressions
The Naída CI M90 is the first Advanced Bionics processor to be produced entirely under the Sonova umbrella from concept on through production. It has the same appearance as the Phonak Naída M90 hearing aid.
The M90 is slightly smaller and thinner than the previous Q series processors, but is now ergonomically shaped for comfort and positioning. The curved shape of the processor and batteries distributes the weight for greater comfort while also ensuring the processor is positioned for optimal microphone placement.
The M Slim HeadPiece now has a permanently-attached cable. This was the design of the older generation Harmony processor’s headpiece and cable, which in my experience made for a robust design that required changing only once during a 7 year period. The design is much thinner and lays flat against the head compared to the Universal HeadPiece design with a detachable cable of the previous Q series. The strain relief features on both ends of the cable have also changed. They are smaller, and may provide improved reliability. Numbered magnets are inserted by your audiologist for just the right amount of grip. If you need a very high-strength magnet, a color cap about the same thickness as the UHP is available. The new Slim AquaMic is exactly the same size as the Slim Headpiece.


The power-up time is a few seconds with only a short start-up jingle. You can miss the chimes if you don’t place the processor on your head fast enough.
Program change times were previously a common complaint with the Q series. It took several seconds to change programs. Savvy users learned to cycle through programs without waiting for each program to come up to speed the process up, but still required sitting in silence until the switch was made. Program changes are immediate unless switching between program strategies (i.e. S or P) in which case the cut out is extremely brief. With AutoSense (described below) manual program changes are largely unnecessary.
Microphones
As with the Q series; the M90 has 4 microphones to choose from: the M T-Mic, the BTE Mic, rear-facing mic, and the headpiece mic. What’s changed about the microphones on the Marvel is the headpiece mic is no longer an option on the 3.5” and 4.25” lengths intended for on-ear wear. A Slim Headpiece with a microphone and a 12″cable length is available for off-ear use.
Longer lengths require the use of the Waterproof Battery, which is ideal for physical activity and water-use to secure the processor to the body. The Waterproof Battery is a small waterproof case that holds the M90 without its T-mic or earhook. It includes a USB-chargeable internal battery. The Aqua Slim HP is a waterproof combination headpiece and waterproof microphone, and comes in lengths of 12”, 18”, 24”, and 42”.
Sound Processing
DR: The M90 is an entirely new processor and system. It has a new sound that is distinctive from the previous Q series. It’s difficult to quantify that difference other than it seems to be more sensitive and offer subtle details that were not there previously both in everyday sounds and music. Adapting to the new sound is effortless and should only offer performance gains.
HS: The sound quality is brighter than with my previous Q70s, even when running the same fixed program. Subtle environmental sounds, such as the collar of my shirt rubbing against my neck. The additional sounds don’t interfere with my speech comprehension. If anything, they make me feel more connected to the environment.
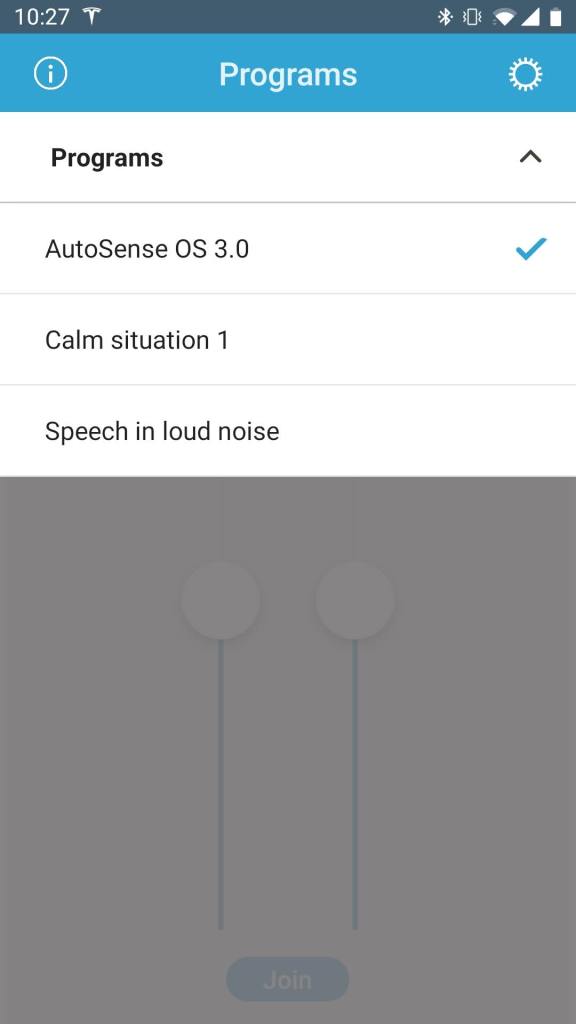
Programs are based on “scene classifiers.” AutoSense uses scene classifiers to identify optimal settings for each acoustic environment you are in. They are: Calm Situation, Speech in Noise, Speech in Loud Noise, Speech in Car, Comfort in Noise, Comfort in Echo, and Music. Four of these, Calm Situation, Comfort in Noise, Speech in Noise, and Comfort in Echo, are blended depending on the environment, rather than having only one selected automatically or manually. In addition to these classifiers, SoundRelax and WindBlock are available in varying degrees of strength that can be set by your Audiologist and are enabled by default. To create a custom program, your Audiologist will select from these scenes to best match the desired results. A recipient desiring a program that is non-reactive to any environment (or no features) would choose “Calm Situation” for that custom program. If a manual beam-former program is desired then the Audiologist would go with “Speech in Noise.”
The Music scene classifier applies a higher input limit and wider dynamic range than other scenes. While conversing in a car, music would play quietly underneath speech, but would become louder and clearer when speech stopped. The compression ratio is slower with this scene. When listening to input over the Bluetooth connection, AutoSense chooses either Media Speech or Media Music, applying similar parameters to optimize the quality of both. A separate custom program may be set for Music is desired.
AutoSense 3.0 is interwoven into the system. It is designed to be the only program you will ever need. Rather than the 5-program structure of the Q series, the M90 offers AutoSense 3.0 plus the option to add up to 4 custom programs. While AutoSense is intended to be the default program upon start-up, it can be placed at the end of your custom program selections and a custom program may be selected as your default. It’s important to understand this structure to avoid confusion. AutoSense has a distinctive audible chime that identifies it as the current program. Any custom programs are identified by the number of chimes, regardless of whether AutoSense is made the default or placed at the end of the custom programs.
AutoSense 3.0 is both the evolution of Auto-UltraZoom as found on the Q90 and is not Auto-UltraZoom at all. It is constantly analyzing your environment and applying changes at rapid fire pace while seamlessly blending the scene classifiers mentioned previously. It all happens without you noticing other than your ability to hear is optimized. In many ways, it is similar to the ClearVoice programming strategy (with which it can be combined), but without the changes in sound quality since it is utilizing front-end features via the microphone.
DR: AutoSense is more of a “challenging environments” program for my use. I can count on it to handle truly loud and challenging environments by focusing on those I need to or cancelling out echo, but it also can overstep in environments I do not consider to be noisy. For the latter, I retain my custom “no-features, wide open IDR” program as a baseline for my hearing. I no longer have a manual UltraZoom program as AutoSense has replaced the need. This is notable since I have never been one to use auto-programs such as ClearVoice or Auto-UltraZoom. UltraZoom was the first truly helpful feature for hearing in noise and was the only program I switched to on the Q70. I am able to remain in AutoSense most of the time with it working to be helpful rather than impede my listening, so I’m finally able to enjoy “Auto-UltraZoom” functionality in advanced form.
HS: AutoSense is my default program, and it is doing a great job in the various environments I’ve encountered to date. My prior default program used 100% T-mic for everyday situations. It’s available in a spare program slot just in case AutoSense doesn’t live up to my expectations. There is one more fixed program – Speech in Loud Noise. So far I haven’t had to switch to either fixed program – AutoSense is doing a great job! I may end up using a fixed program when I’m only listening to music through the microphones.
Bilateral Features
The Q70 has two microphones at the top of the processor. They work together in UltraZoom to focus the sound directly in front of you. This works great in a noisy restaurant.
The Q90 adds StereoZoom for bilateral and bimodal users. It uses mics from both processors, which communicate with each other wirelessly, to do an even better job focusing on the sound in front of you. The greater separation between mics facilitates this advanced capability. Bimodal users can take advantage of this with the Link hearing aid. This is one example of how Advanced Bionics processors and Phonak hearing aids work together. No other cochlear implant and hearing aid combination works like this.

For bimodal users, the Phonak Naida Link M Hearing Aid includes all of the bilateral features when you have one cochlear implant and one hearing aid. It’s available as an option on the M90 order form.
Speech in 360 is a separate custom program that builds on Speech in Loud Noise. It focuses on sounds in front of you. But if a waiter starts talking to your side, it shifts the focus to that direction! It is not part of AutoSense, so you have to switch to that program. I plan to replace my custom Speech in Loud Noise program to Speech in 360. AutoSense does a great job of deciding when to use Speech in Loud Noise, so it isn’t needed as a separate program.
As with the Q70 and Q90, you can change programs or the volume of both devices by pressing a button on either one. If AutoSense continues to perform as well as it has been, there will be no need to press buttons to adjust anything about your hearing.
The Q70 and Q90 are the only cochlear implant processors that can be programmed for both ears. You can put either processor on either ear, and the correct set of programs will load. This is useful for bilateral users in case one processor has a problem. If you prefer to use one side more than the other, or perform better on one side, you can just put the remaining processor on your best side. In addition, you can swap sides as part of a debugging process to localize the problem. This capability has been dropped from the M90 processors to make room for the more advanced features.
Wireless Features
The first wireless feature you will encounter is the wireless programming interface at the audiologist’s office! There are no cables from the audiologist’s computer to your processor(s). The audiologist can even program the M90 processor on one ear, and the corresponding Link hearing aid on the other side, both wirelessly, at the same time. All changes the audiologist makes are updated right away. You can hear for the entire programming session. When all settings are finalized, it only takes a few seconds to save them to the processor.
The M90 is the only CI processor with true Bluetooth connectivity to all devices! It even works in true stereo for bilateral and bimodal users. All other CI processors use Made For iPhone and/or ASHA (Android 10 and up) connectivity for hearing aids. Those devices are only able stream from mobile devices with the correct OS. They are also only one-way audio connections – you need to use the mic on the phone to talk, so the phone can’t be in your pocket or purse. Some other CI processors do have full Bluetooth connectivity – but only with an intermediary device that you clip onto your clothing or wear on a neckloop.
The Marvel hearing aids and CI processor connect via Bluetooth as “headphone” or “headset” to all Bluetooth devices with Bluetooth version 2.1 and above. They even work with many older phones utilizing versions of Bluetooth below 2.1! The M90 can be a complete wireless headset for video calls on any Bluetooth device, including your computer.
The connection strength of the M90’s Bluetooth is excellent and comparable to high-end consumer Bluetooth headphones. There is never a break in the stream, so you can move one or two rooms away from your phone without losing connection. The stream is in true stereo when listening to music or media, and will also stream to both sides on phone calls for bilateral or bimodal users with the Phonak Naída Link M.
Having true Bluetooth on-board the M90 gives you the ability to make hands-free phone calls with the phone in your pocket or purse. You can answer a call or hang up with a press of a button on the processor. Calls are streamed directly to your processor/s with your own voice picked up by the BTE mic. Call clarity is pristine on both ends with your out-going voice sounding as clear to those on the other end as if you were holding the phone up to your mouth. Having had a very social weekend to celebrate my recent birthday, I found myself being the one in the group (everybody else with normal hearing) to make calls to various restaurants to gather information while there was lots of noise on both ends of the conversation. Not a single word was missed nor was there any struggle at all! During a call, you can adjust the volume either with the M90 volume buttons or on your phone’s volume rocker.
Up to two devices can be paired and held in memory on the M90. Only one of these devices can be connected to the M90 at a time. When switching between the two devices that are both in range and powered on; you need to first disconnect one (not “forget” or unpair) and then connect the other device (usually done within the Bluetooth settings for that device). Switching may also happen automatically where the connected device is turned off and the other device then resumes connection. Additional pairings are possible, but if two devices have already been paired, one device will be replaced by the new device. You will need to re-pair the previously paired device to restore it to memory. Presently, my smartphone and my PC are the two paired devices I can switch back and forth from with little fuss.
If your phone is paired to your car audio system and to your M90 processor, phone calls will go to the processor when you are not in the car. The car takes priority, so calls automatically go through the car audio system when you are in the vehicle. If you prefer to use the Bluetooth connection to the M90 instead of the car audio system, don’t pair the phone with the car.
The TV Connector accessory adds further connectivity options into the mix. It utilizes Airstream; Phonak’s proprietary streaming protocol. This is different from Bluetooth in that multiple users can connect to one TV Connector device to hear in full audio quality stereo sound on demand. Despite the name, TV Connector can be used with any device outputting sound via either TOSLink/Optical or 3.5 mm analog (ideal for headphone outputs.) You can plug it into the headphone output jack of a computer as a method to simplify switching between Bluetooth streaming from your phone and the computer. Airstream is a stereo stream that does not merge into mono for unilateral recipients (Bluetooth will merge as needed.)
Other wireless accessories include the Phonak Roger Select IN, the Phonak PartnerMic, and the Phonak Remote Control.
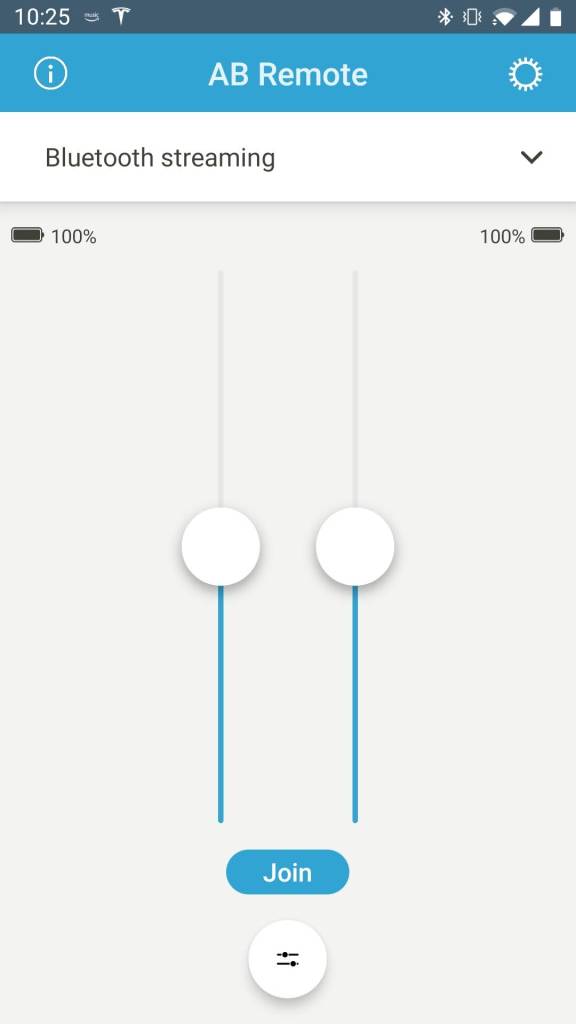
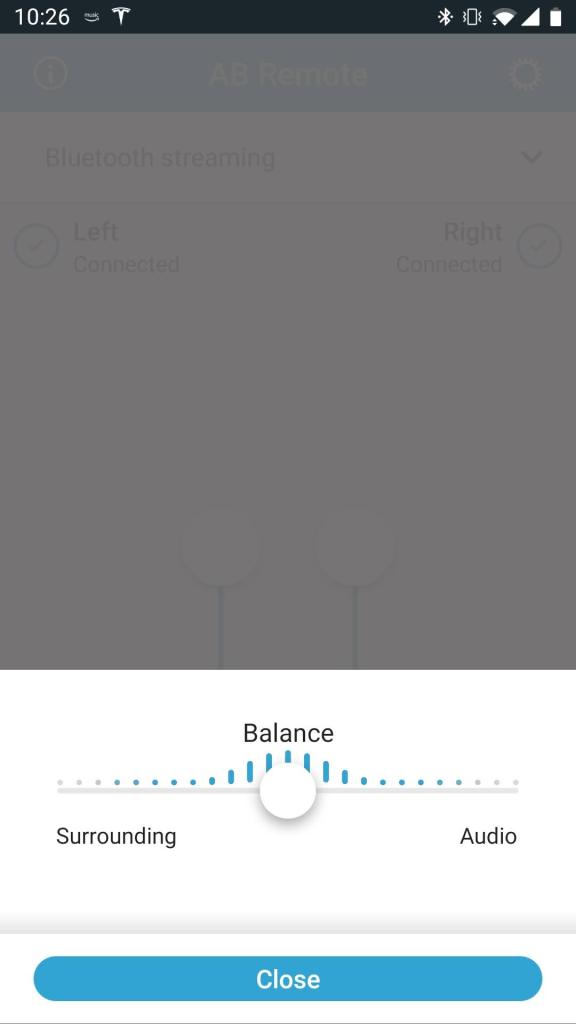
AB Remote App
The AB Remote app, available for Android and iOS, gives you access to some basic features from your phone. You can check the battery levels, adjust volume (independently or both at once), set surround mix, and change programs from a drop-down list. In addition, you can check the status of the processor, play a beep, or blink an LED, which is useful for caregivers. The app may be downloaded from the App Store or Play Store and utilized in demo mode for those wishing to check it out in advance.
Batteries
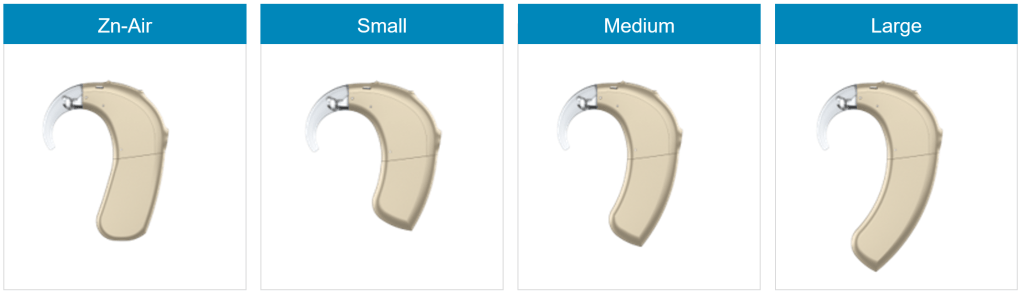
Each of the three rechargeable battery sizes is smaller than the corresponding options for the Q series. A case for disposable Zinc-Air batteries is also available. I got 21 hours of battery life with new 150mAh medium batteries on the M90. With new 170mAh batteries on the Q70, I got about 16 hours. This means the combination of the new processor and software is about 50% more efficient!
Waterproof Battery

The M90 can be put inside the new M Waterproof Battery that essentially turns it into a smaller Neptune (the world’s only fully waterproof CI processor, which has recently been discontinued) with wireless capabilities. The casing has a battery built-in and is charged directly via micro-USB. You could theoretically install a backup processor if you have one, leave the case locked, and use it as a fulltime off-ear processor for physical and water activity. The form factor ensures the processor is protected and secured to your body. Ocean waves will knock a CI processor right off your head without fail, so if it is not secured it will turn up on a deserted island someday.
I primarily use this during my weight-lifting workouts. I can get drenched with sweat without worry for the processor. The wireless streaming lets me listen to my music playlists to power me through my routine. I’m looking forward to wearing it at amusement parks while riding roller coasters knowing it is secure and enjoying hearing the terrified screams of fellow riders.
Charger

The battery charger is comparable in size to the Q series charger. However, it only charges three batteries at a time, while the Q charger has four slots. As a bilateral user, I like to have four batteries on the charger overnight. Two go on my processors in the morning, and the other two are carried with me in case the batteries die while I’m out for the day. Now I need to keep two chargers on my nightstand to charge four batteries at once. Update: With 21 hours on a medium battery, you may not worry about a battery dying during the day. You can still carry spares in case of emergency, but they don’t have to be charged every day.
The LEDs are much dimmer than those on the Q series charger. This is a welcome improvement for people who like to sleep in a very dark room.
The charger uses a Micro-USB connection. The supplied cables are pretty stiff. Aftermarket quality flexible cables will make the wire routing for the two chargers as easy as possible.
Dryer

The Cedis dryer supplied with the kit is much smaller than the Zephyr which came with the Q series. Two M90 processors with headpieces can fit in the compartment without undue flexing of the headpiece cable. The charger uses a mini (not micro) USB connector. While the dryer and battery chargers usually remain plugged in, so you don’t have to fiddle with micro and mini USB connectors on a regular basis, a system where all the accessories use USB-C would be welcome.
Summary
The M90 provides enhanced sound quality and a completely natural user experience. With AutoSense, most users will find no need to switch programs at all. The smaller size, lighter weight, and ergonomic shape result in a more comfortable and secure fit.
Connectivity to any Bluetooth device is awesome! Hands-free phone calls and video conferencing provides crystal-clear sound on both ends of the conversation.
Pros:
- Smaller, lighter, more ergonomic
- Much better battery life
- AutoSense automatically adjusts program settings for all listening environments
- Improved sound quality – clarity and brightness, more environmental sounds
- Full Bluetooth connectivity to any device
- Hands-free calling and teleconferencing
Cons:
- Each processor must be assigned to one ear
- The battery charger can hold only three batteries
- Mini-USB and Micro-USB connectors on the charger and dryer. Stiff cables.
About the Authors
David Ryan is a music and tech enthusiast as well as a 15-year unilateral implant recipient after 25 years of hearing aids. The loss of music and social communication in his early 30s motivated taking the next leap into the world of bionic hearing. Those hopes and dreams have been fully realized via successive advances in internal and external tech.
Howard Samuels is a late-deafened adult who received his first implant in 2005 and his second in 2007. He’s progressed through several generations of processors and sound processing strategies, and has been enjoying better hearing with each successive generation.
MED-EL USA Releases New S-Vector Magnet for SYNCHRONY 2 Cochlear Implants
Editor’s note: MED-EL announces a stronger magnet today in the implant, which provides improved ability for single-piece processors like the RONDO 3 to remain in place for active situations.

MED-EL leads the hearing industry with stronger magnetic attraction and optimized retention with the same outstanding MRI features and warranty protection
March 16, 2021 – (DURHAM, NC) – In 2014, MED-EL forever changed the hearing implant industry with the launch of SYNCHRONY, the world’s first cochlear implant featuring a patented diametric magnet approved for 3.0T MRIs without requiring magnet removal. MED-EL is taking its innovative magnet design to the next level by releasing an all-new diametric magnet with S-Vector technology. The U.S. Food and Drug Administration (FDA)-approved S-Vector magnet is a feature of SYNCHRONY 2 cochlear implants that ship starting today.
The unique S-Vector magnet for SYNCHRONY 2 is 25% stronger than the first-generation magnet. A stronger magnet is ideal for active recipients with off-the-ear single-unit audio processors like RONDO 3. Despite increased magnetic strength, there are no changes to existing MRI scanning instructions. The S-Vector magnet also has the same small MRI artifact as the first-generation magnet.
MRI scans with the S-Vector magnet are covered by MED-EL’s MRI Guarantee. MED-EL is the only manufacturer to offer its users a comprehensive global lifetime MRI guarantee ensuring that the magnet stays in place during an MRI scan and is not damaged, nor does it dislocate or demagnetize.*
The S-Vector magnet is now available in SYNCHRONY 2 implants that feature MED-EL‘s FLEX electrode portfolio.** Engineered to protect cochlear structural integrity, MED-EL’s atraumatic FLEX electrode arrays feature ultra-flexible, wave-shaped wires and a tapered tip for reduced rigidity. The large selection of FLEX electrodes helps ensure each recipient receives the best possible array to fit their unique cochlear anatomy.
“Over the past 25 years, we have continuously developed cutting-edge magnet designs to tackle the issue of MRI compatibility and hearing implants for our recipients,” said Raymond Gamble, President & CEO, MED-EL North America. “We’re thrilled to announce that we’ve further advanced our magnet design and have now made the second generation available to the U.S. market.”
“A stronger implant magnet results in a stronger holding force, providing outstanding flexibility in finding the ideal fit with the user‘s audio processor,” said Martin Zimmerling, Head of Implant Development at MED-EL. “The S-Vector provides more holding force and provides a great option for adapting to the user’s individual needs. This gives our recipients peace of mind knowing their audio processors will stay securely in place while benefiting from the industry-leading MRI features for which MED-EL’s cochlear implants are known around the world.”
Advanced Bionics M90 Unboxing Pictures
Gallery

This gallery contains 10 photos.
Cochlear Kanso 2 Processor Review
Iraida Franceschi

As a child, I had allergic reactions to everything and anything that grows under the sun, (i.e., grass and tree pollen, dust, mold, etc.) So I commonly experienced recurrent ear infections. Therefore, I was always given antibiotics or antihistamines, and ironically, it was also said that the medications I was taking affected my hearing loss as well. Either way, I was one of those people that thought hearing-aids were always going to be part of my life. Never imagined that hearing-aids were no longer going to be sufficient for me. Yet, during a hearing evaluation, my audiologist told me I should consider cochlear implants, and I went home feeling overwhelmed. Fast forward to 2011, and after being told I qualified, I started to research more and more about cochlear implants all the while asking questions in social media. It was all a little daunting but having most of my worrisome questions answered by my doctor and by other cochlear recipients slowly gave me confidence to go forward with the process.

In 2012, I decided to get a cochlear implant – and it was a success! Having had such a great experience with my first, I decided to go for my second a year later, becoming bilateral in 2013. At that time, I received the Nucleus 5 processors (for both sides), and I continued enjoying the journey for approximately 8-9 years until it was time to think about an upgrade. Cochlear Americas had just released their newest processor: Kanso 2. So, I knew that all I needed to do to get the upgrade process started was to email or call Cochlear, as they now have a Reimbursement and Insurance Services department to help with the process. Cochlear Americas also takes care of contacting my Audiologist and ENT Surgeon to obtain the letter of medical necessity and other information pertaining to the upgrade. Consequently, I am happy to announce that my upgrade was successfully approved, and that I have had the Kanso 2’s since November 2020.

Key Features of the Kanso 2
Built-in Rechargeable Battery: The rechargeable battery is non-removable, and internally settled (built-in) inside the Kanso 2. This means I do not need to worry about putting on or taking off the rechargeable battery, because the Kanso 2 is a one-piece unit processor. This is also known as an Off-The-Ear (OTE) processor.
Easy on and off functionality: I can turn it on by placing it on my implant and turn it off by taking it off. Or I can tap twice to turn on and tap three times to turn it off while it’s still on my implant.
SmartSound iQ with Scan Technology: The scan technology is automatically listening to the surrounding sounds, and it adapts or changes the settings depending on the environment. There are six scenes in total: Quiet, Speech, Speech in Noise, Noise, Music, and Wind.
Direct connectivity to a smartphone: Connecting to a smartphone (Android or iPhone) to stream and enjoy music, movies, videos, phone calls and video calls.
Nucleus Smart App: The app can help control the sound settings, track hearing data, and battery level. It is free and can be downloaded from the Google Play Store or Apple Store.
Dual microphones: This is important to help filter out background noise and enhance the hearing experience.
Once I received the processors, I called my audiologist to make a mapping appointment. This was necessary because the Kanso 2’s I had received are the latest and newest processors available – and were completely different from the processors that I was using.
In comparison, the Nucleus 5 is a behind the ear (BTE) processor, whereas the Kanso 2 is an off the ear (OTE) processor. This allowed me to free my ear for wearing glasses or sunglasses — and masks which are required now due to the pandemic.
With an Off-the-Ear processor, I won’t need to worry about the coil or the cable, as the Kanso 2 is a one-piece unit. Moreover, the Kanso 2 has a built-in rechargeable battery in it that is non-removable. This means that when I am done for the day, I can simply place my processors in the charger/dryer box and close the lid to allow it to automatically begin charging wirelessly. It takes approximately 3 to 4 hours to charge in full. However, because most of us sleep 6-8 hours at night, it has been said we can leave them safely drying overnight, the technology is smart, and the charger/dryer won’t overcharge the processors.
Once I go about my daily routine, and if I am attending online meetings that day, I know I can use the Mini-Mic 2+ to stream directly into the Kanso 2’s. All I have to do is plug one end of an audio cable into the headset port on my computer, and the other into the Mini Mic 2+. Then I select the audio input on the Mini Mic 2+ by pressing its mode-selector button three times to set it to the music icon. The Mini Mic 2+ streams sound wirelessly to both Kanso 2 processors, and its mic picks up my voice. This, to me, is a game changer! The sound quality is impressive, and I can follow conversations with my colleagues via Zoom. Just to be able to listen and chime in with constructive feedback, is all worth it.

I am also excited to share that working out with my Kanso 2’s has been easier. Plus, I found out that the Kanso 2’s have a water resistance rating of IP68, which means that the sound processor can withstand sweat much more than my Nucleus 5’s did as these had an IP57 rating. I could also get rained on or splashed and not have to worry about damaging my Kanso 2’s. In fact, I have been reading about the IP68 rating, and it has been said that the Kanso 2’s can be submerged underwater (to a maximum depth of 1.5m) for up to thirty minutes without any damage.
After I am done teleworking, I am usually using my iPhone, listening to music, or browsing TED Talks videos to listen to as that is now part of my daily audio rehab. With the iPhone, I don’t need any other intermediary devices, as the Kanso 2’s are able to stream sound directly to my implants. As a matter of fact, this is probably the easiest thing to do, to make calls or receive FaceTime calls from my daughters, because once the iPhone is paired, I really do not need to press any buttons. It just rings and streams without delay.
I can scroll down the timeline in social media and if I click on a video, it will instantly stream the sound to my Kanso 2’s. If I get interrupted with a call or a FaceTime call from my daughter, the call will take over and instantly connect. Then when I hang up, the video that I was watching resumes playing. It surely is as easy as I am making it sound!

The Kanso 2’s can also be used with the Nucleus Smart App, which is a free app that can be downloaded from the Apple Store – or Google Play (for Android). Once downloaded and paired, I can check my settings, such as volume, sensitivity, treble and bass – or I can change programs or leave it on Scan, which is a special program that listens to the surrounding sounds near me, making adjustments depending on whether there is noise or quiet, speech, music or even wind.
There is also a Hearing Tracker feature, which displays Time in Speech (per day) in which I can tell it to “set a goal” depending on the number of hours I wish to achieve and it will highlight the days I have met the goal. The other feature in the Hearing Tracker is the Coil-Offs for tracking the number of times the coil was disconnected from the implant each day.
If I ever misplace my Kanso 2’s, I can tap the “Find my Processor” button to show me the location of my Kanso 2’s to help me find them. So, there are quite a lot of features in the app that are useful. But the feature I use the most is the “status” of the batteries, as the Nucleus Smart App can display the amount of charging power remaining in the rechargeables, and it does so by displaying the percentage. This is a very convenient feature.
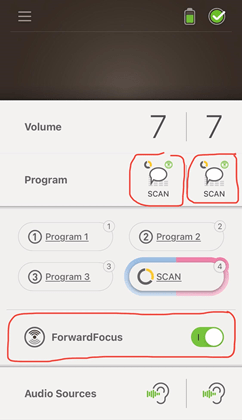
I also tested the “Forward Focus” feature, which is also a very effective setting for when I wish to drown out any noise behind me. So, I placed my back behind the TV, which was playing really loudly, and I had told my spouse to read a paragraph or two out of a book I had grabbed. He read out loud and I started to notice that his voice was in focus while the TV noise was reduced to some degree – but noticeable enough for me to enjoy this feature.
Now, in regards to the Kanso 2’s SmartSound iQ with Scan Technology, I was surprised that I didn’t notice much difference. I have tested this program by playing loud music from my Amazon Echo to make the Scan recognize music and change settings, and it certainly did so. Then I would walk away to the dining room and start a conversation with my husband, and the Scan program would automatically change to the Speech setting. All these changes were happening as I expected them to, but in a very subtle and unnoticeable way. In fact, the only way I could tell it was changing was because I was looking at the Nucleus Smart app and watching the changes. Perhaps I should give more time to the Scan feature or bring it to the attention of my Audiologist for enhancement.
Now let me bring up a different aspect about the built-in rechargeables. As mentioned, the rechargeable battery is part of the Kanso 2 — which brought up concerns (to me) about whether or not it would last me all day long. This is because I didn’t want to have to take off my Kanso 2’s in the middle of the day, or during a meeting, or even late at night while watching a movie. The difference is that the Kanso 2 would need to be removed to be charged, and I would be left without sound. However, after doing all the testing, I have come to realize that I am able to wear my processors for 2 days without charging them every single night. That’s how much power there is in the Kanso 2’s (with my Scan and Mapping programs). In the case of a power outage or when going camping, I can use the Portable Charger that was included in the upgrade kit. The portable charger will charge similar to the charger/dryer unit for approximately 3-4 hours, which would give me another 2 days to wear my Kanso 2’s. If, however, I were to go camping for a whole week, or if the power outage were to last much longer, my back up plan is to use my AC outlet portable power station. This can be purchased online and is available at outdoors sporting stores.
Either way, I can tell that the new Kanso 2’s are going to be my new connection to life. I plan to explore the new features and enjoy the new experiences my Kanso 2’s bring me. After all, this is the technology I have always dreamed of having, and I couldn’t feel any more grateful to be able to do so.
About the Author

Iraida Franceschi is a boricua wife, mother of two adult daughters, and one of the five Admins of Cochlear Town USA, a closed-group on Facebook. She mentors and corresponds with other candidates during her spare time.
MED-EL Announces Global MRI Guarantee for Cochlear Implants
Globally unique MRI Guarantee: the highest MRI safety with hearing implants from MED-EL
Worry-free, safe magnetic resonance imaging (MRI) with MED-EL cochlear, middle ear or bone conduction implants – now with lifetime guarantee! For immediate access to an MRI examination without surgery, discomfort or hearing downtime.
Innsbruck, 02.02.2021: The innovative design of the implant magnets from MED-EL is completely safe during an MRI examination. As of now, the global innovation leader in hearing solutions is emphasizing this safety with an MRI guarantee that is as yet unmatched. With this, MED-EL is the only manufacturer to accept liability for damage to all cochlear implants that have been produced since 1994, all bone conduction implants and the current generation of middle ear implants during an MRI examination. Users of these MED-EL implants can therefore not only undergo an MRI examination at any time without the need for surgical removal of the magnets* and without pain – they can also do so safely and with no worries.
Three in four people will require an MRI scan in the next ten years**. Whether routine or in an emergency: patients with severe hearing loss may face a challenge if the magnets in their hearing implants are not safe inside the MRI machine. This requires the prior surgical removal of the magnets. Hearing ability is temporarily lost and long-term damage to the implant may occur. If the patient declines surgery, they may suffer from severe pain during the examination coupled with the possible dislocation of the magnet.
The highest MRI safety with MED-EL
Not all implants carry this risk. As a leading manufacturer of implantable hearing solutions, MED-EL has for more than twenty years made a point of only producing implants that offer outstanding MRI safety. To emphasise this requirement, MED-EL offers the unique MRI Guarantee which means no damage to the device during an MRI examination. MED-EL is thereby the only manufacturer to issue its users with a comprehensive global lifetime MRI guarantee.
“The MRI Guarantee ensures more safety, comfort and peace of mind during MRI examinations,” says Alexander Hofer, Corporate Director of Product Management at MED-EL. “We take care of our users for a lifetime and they should have the opportunity to make use of everything modern medicine has to offer. This is especially important when diagnosing serious illnesses such as cancer, tumours, multiple sclerosis or Parkinson’s disease,” stresses Alexander Hofer.
Being able to undergo magnetic resonance imaging immediately can also be crucial in emergencies such as an accident. A further benefit: people with severe hearing loss only have to go without their ability to hear during the MRI examination itself.
Innovative technology excludes complications
The safe and reliable technology from MED-EL ensures that the magnets in place during the magnetic resonance imaging are neither damaged, nor move causing pain. Only the external components, such as the audio processor, must be removed prior to the MRI examination.
“MED-EL has developed its own highly innovative, patented cochlear implant system with state-of-the-art implant magnets and their fixation within the implant. This excludes, for example, a dislocation of the magnet during an MRI scan, which can be very painful for the patient,” says Martin Zimmerling, Head of Implant Development at MED-EL. “With the SYNCHRONY cochlear implants, the magnet can freely rotate and align according to the magnetic field of the MRI scan. With the altered implant design, undesirable forces during the MRI scan and the associated pain for the patient are reliably prevented,” explains Martin Zimmerling. “We are proud not only that the latest generation of our cochlear implants is compatible with MRI machines, but that all cochlear implants produced since 1994 are too, as well as all bone conduction implants and the latest middle ear implant from MED-EL.”
The increasing significance of magnetic resonance imaging makes it clear how important it is to provide people with severe hearing loss access to this gentle and high-resolution imaging process. MED-EL rises to this responsibility and provides extra security and comfort for all users with the MRI Guarantee.
*So as to avoid imaging difficulties in the immediate vicinity of the implant, it may be advisable to remove the magnets.
**Source: OECD. Magnetic resonance imaging exams 2016–2019.
Editor’s note: Current implants from Advanced Bionics, Cochlear, and MED-EL are all tolerant of 1.5T and 3.0T MRIs with the magnet in place. Also, Advanced Bionics’ previous implant, the HiRes 90k Ultra, is tolerant of 1.5T MRIs with the magnet in place.